3 Types of Heat Transfer: An Overview to Energy Efficiency
By HeyHome • October 30, 2023

The sensation of a hot cup warming our hands on a cold day, the comforting feeling of a cool breeze on a summer afternoon, and the touch of sunlight on our skin — all these familiar experiences are underpinned by a fundamental process: heat transfer. Yet, the concept of heat transfer is much more than mere experiences of warmth or cold; it is a pivotal factor influencing our daily lives in profound ways.
In the world of physics, heat transfer represents the transfer of heat or thermal energy between objects, or within an object. There are three principal mechanisms through which this transfer occurs, shaping the experiences and technologies we interact with daily. These are:
- Conduction: The process of heat transfer through direct contact, such as when the warmth from a hot beverage seeps into our hands.
- Convection: A mechanism where heat transfer occurs through the bulk motion of fluids, like the circulating hot air that warms a room.
- Radiation: Heat transfer in the form of electromagnetic waves, such as the infrared radiation that we feel from the Sun, even without physical contact.
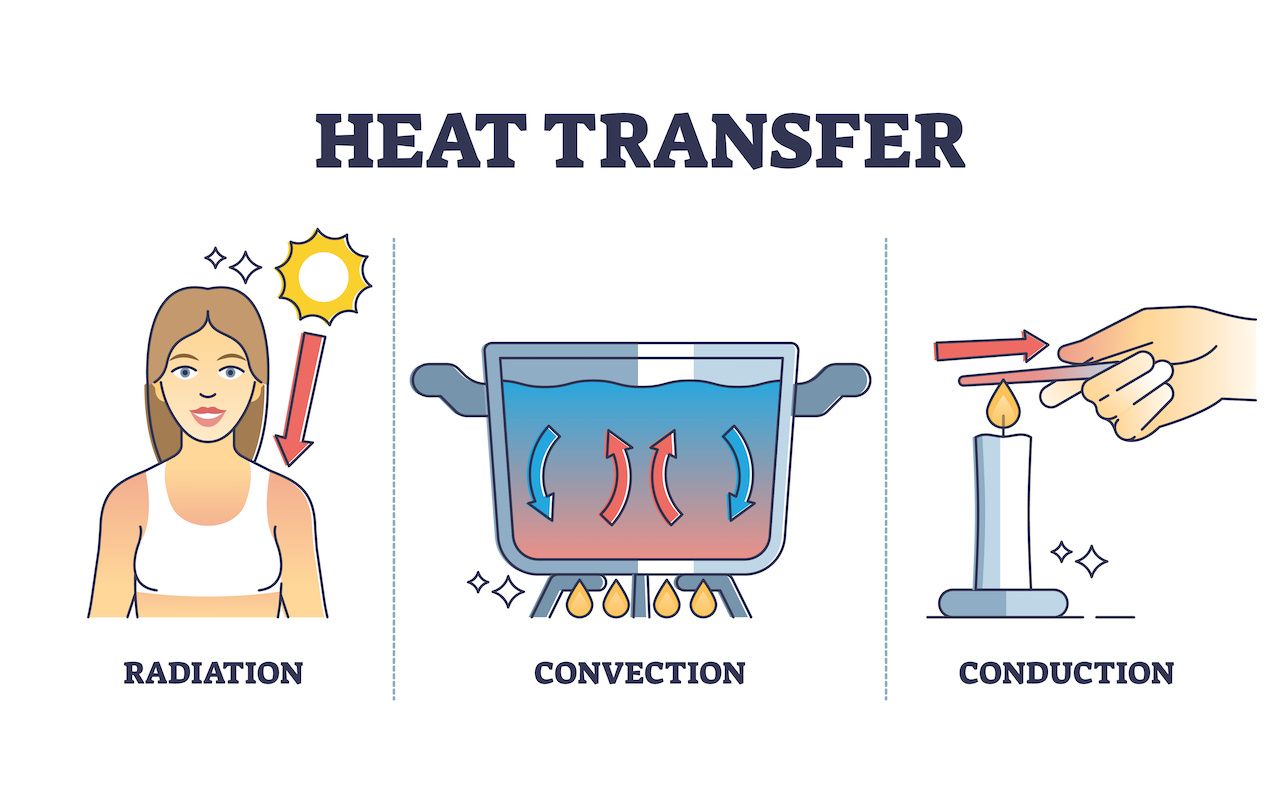
Recognizing and understanding these 3 types of heat transfer is essential. Not only do they dictate our sensory experiences, but they also underlie myriad technologies, from energy-efficient windows in our homes to the heat exchangers in industrial settings.
A grasp on how heat is transferred, the factors influencing it like temperature difference, and the tools to manipulate it, empowers us to navigate our world more efficiently and sustainably. As we delve deeper into this topic, prepare to view the world around you with a renewed perspective, seeing the hidden dance of energy that constantly unfolds.
Conduction: When Objects Meet
If you've ever grasped the handle of a metal spoon left in a hot pot or felt the chill of a tile floor on a winter morning, you've experienced conduction firsthand. At its core, conduction represents the process of heat transfer through direct contact between substances without any bulk movement of the material itself. But what's happening on a microscopic level?
The Fundamentals of Conduction
Heat, or thermal energy, is essentially the kinetic energy of atoms and molecules vibrating in a material. In solids, these atoms and molecules are closely packed. When one part of an object is heated, the particles in that region start vibrating with greater intensity. These vibrations then spread to neighboring particles, transferring the heat. This process continues until the heat has been uniformly distributed or until a temperature difference no longer exists.
In the realm of conduction, two objects or regions don't just passively share their heat. The rate and efficiency at which this heat transferred occur hinge significantly on the properties of the materials involved.
Join our newsletter
Stay on top of the latest in landscaping and lawn care with one valuable tip right in your inbox every Saturday morning.
Everyday Encounters with Conduction
To illustrate, let's consider a few commonplace examples:
- Touching a Hot Cup: When you grasp a hot cup of tea, the heat from the cup is conducted through the solid material of the cup into your hand. This happens because the particles in the hotter region (the cup) pass on their energy to the particles in the cooler region (your hand).
- Solid Materials Conducting Heat: Metals, for instance, are excellent conductors. If you've placed a metal rod with one end in a flame, you'd quickly feel the other end becoming hot. The metal's free electrons facilitate the swift transfer of heat, making it an efficient conduit for thermal energy.

The Role of Thermal Conductivity
While some materials, like metals, readily conduct heat, others, such as wood or rubber, resist heat transfer—these are insulators. This propensity to conduct or resist heat is quantified by a property called thermal conductivity. The higher the thermal conductivity of a material, the more efficient it is at conducting heat.
Thermal conductivity doesn't just dictate our day-to-day experiences; it has broader implications for energy efficiency. For instance, in home construction, materials with low thermal conductivity are often chosen as insulating materials, ensuring homes remain warm during winters and cool during summers without excessive energy consumption. Such choices directly impact energy bills and the overall carbon footprint of a residence.
Additionally, understanding thermal conductivity aids in designing efficient heat exchangers in industrial setups, optimizing the transfer of heat without waste.
In essence, conduction is a silent player in our daily lives, influencing comfort, energy consumption, and technological processes. As we move forward, it's essential to recognize how this fundamental heat transfer mechanism intertwines with its counterparts, convection and radiation, shaping our world in myriad subtle ways.
Convection: Motion Brings Change
Convection paints a vivid image of swirling liquids, rising steam, and the comforting warmth that envelops you when you step into a heated room. Unlike conduction, where heat is transferred through stationary objects, convection revolves around the dynamic movement of fluids (liquids and gases) and the integral role this motion plays in distributing thermal energy.
The Dynamics of Convection
At the heart of convection lies the principle that fluids change their density as they're heated or cooled. Consider a pot of water set to boil. As the water at the bottom heats up, it expands and becomes less dense than the cooler water above. Being lighter, the hot water rises, and in its place, the cooler water sinks to the bottom to get heated. This cyclical process results in the formation of convection currents, which are instrumental in transferring heat throughout the fluid.
Natural vs. Forced Convection
Convection can manifest in two primary forms:
- Natural Convection: This occurs purely due to temperature differences within the fluid. In the absence of any external force, the fluid motion arises from the buoyant forces resulting from density variations. A classic example is the warm air rising near a heater while cooler air descends to take its place.
- Forced Convection: Here, an external agent, like a fan or a pump, propels the fluid, ensuring the heat is dispersed more rapidly and uniformly. Your car's cooling system, which uses a pump to circulate the coolant, is an instance of forced convection in action.
The Crucial Role of Temperature Difference
Central to the onset of convection is the temperature difference that exists within the fluid. This difference is the driving force that sets the fluid in motion. The greater the temperature gradient, the more pronounced the convection currents. It's this gradient that determines the speed and efficiency at which heat is dispersed throughout the fluid.
Everyday Encounters with Convection
Convection isn't just limited to pots of boiling water or room heaters; it's deeply embedded in our daily experiences and even within our bodies. Here are a few instances:
- Blood Circulation: Our circulatory system is a marvel of natural convection. As blood circulates, it helps regulate our body temperature, ensuring a consistent warmth is maintained throughout.
- Water Boiling: As touched upon earlier, the dance of hot water rising and cooler water sinking in a boiling pot is a classic display of natural convection.
- Hot and Cooler Water Interactions: Imagine pouring cold water into a tub of hot water. The ensuing motion, where hot water rises to the surface while the cooler water sinks, is convection at work, helping in homogenizing the water temperature.
Convection is a testament to nature's ingenuity, ensuring heat transfer isn't just confined to static objects but encompasses the dynamic world of fluids. Whether it's the gust of hot air you feel when opening an oven or the rhythmic flow of blood in your veins, convection, in all its forms, plays a pivotal role in determining how heat shapes our experiences.
Join our newsletter
Stay ahead of the curve in all things outdoor.
Get the inside scoop on the latest landscaping, lawn care, and fencing trends with 1 actionable tip every Saturday morning.
Radiation: The Invisible Messenger of Thermal Energy for Energy Efficiency
As we delve deeper into the world of heat transfer, we encounter radiation, an enigmatic form of energy transfer that doesn't rely on any medium. Unlike its counterparts, conduction and convection, which necessitate material interaction or fluid motion, radiation heat transfer operates on a more ethereal plane. In the vastness of outer space, where there's a stark absence of matter, it's radiation that reigns supreme, bridging the cosmic distances and connecting celestial bodies.
Distinctive Dynamics of Radiation Heat Transfer
The unique characteristic of radiation is that it doesn't require a physical medium to propagate. Whether it's the heat transferred from the sun to Earth or the warmth emanating from a campfire, radiation operates independently of direct contact or fluid movement. This form of heat transfer occurs through electromagnetic radiation, a spectrum of energy waves that includes visible light, ultraviolet light, and infrared radiation.
Electromagnetic Illuminations: Visible Light and Infrared
Our daily encounters with radiation predominantly revolve around two segments of the electromagnetic spectrum:
- Visible Light: The colors we perceive are a result of light waves reflecting, refracting, and interacting with objects. While visible light plays a less direct role in heat transfer, it's integral to our understanding of radiation.
- Infrared Radiation: In the realm of radiation heat transfer, infrared holds a prime position. This is the thermal radiation that objects emit based on their temperature. It's why we feel warmth when near a source of heat, even if we're not in direct contact.

Earth's Radiant Warmth: The Role of Solar Radiation
The sun, a colossal fusion reactor, continually showers the Earth with solar radiation. Upon reaching the earth's surface, some of this radiation is absorbed, warming our planet, while the rest is reflected back into space. This delicate balance ensures Earth remains hospitable. However, the presence of greenhouse gases like carbon dioxide can trap more infrared radiation, leading to phenomena like global warming.
Our Personal Radiator: The Human Body's Mechanism
Interestingly, our own bodies are sources of infrared radiation. Based on our body temperature, we emit infrared rays, which can be detected using thermal cameras. This is why, in cold surroundings, we can feel the heat transferred from another person standing close by, even without physical contact.
In conclusion, radiation is an intriguing mode of heat transfer, beautifully intertwining the realms of the microscopic and the cosmic. It's an omnipresent force, ensuring that even in the emptiness of space or the isolation of a cold winter night, heat finds its way, connecting entities across distances and making its presence indelibly felt.
Real-life Implications: The Power and Efficiency of Heat Transfer
In our day-to-day lives, the intricate dance of heat transfer is omnipresent, orchestrating a symphony of warmth and coolness that touches every moment. Often, the three cardinal types—conduction, convection, and radiation—don't act in isolation; they occur simultaneously, weaving a tapestry of thermal dynamics around us.
Consider a simple scenario: brewing a pot of tea. When you place a kettle on a hot stove:
- Conduction ensures that the heat from the burner is transferred to the base of the kettle, warming the water inside.
- Convection currents circulate this hot water, causing it to rise and cooler water to sink, ensuring uniform heating.
- Radiation from the burner also contributes, as it heats the sides of the kettle.
Such everyday occurrences highlight how understanding heat transfer can lead to more energy-efficient practices. By leveraging the nuances of how heat is transferred, we can design better kitchen appliances, create more effective heating systems, or even optimize our homes with energy efficient windows and insulation.
In essence, a deeper comprehension of heat transfer not only enriches our appreciation of the world around us but also empowers us to harness its potential. As we strive for a sustainable future, such knowledge becomes a beacon, guiding us toward practices that combine comfort, conservation, and efficiency.

Harnessing Heat for a Sustainable Future
Throughout our exploration of the 3 types of heat transfer, we've unearthed a rich tapestry of knowledge that permeates our daily lives. From the warmth of a freshly brewed cup of tea to the sunlight radiating upon the Earth's surface, heat transfer is not just an academic concept—it's a lived experience. Understanding the nuances of conduction, convection, and radiation is not only an intellectual pursuit but a pragmatic one. In a world increasingly focused on energy efficiency and sustainable practices, this knowledge holds the key.
As we move forward, let's not just stop at understanding. Let's dive deeper, apply, and innovate. With the principles of heat transfer as our guide, we can navigate toward a future where we harness heat responsibly, optimizing our surroundings for comfort and sustainability.
Join our newsletter
Stay ahead of the curve in all things outdoor.
Get the inside scoop on the latest landscaping, lawn care, and fencing trends with 1 actionable tip every Saturday morning.